Eye Drops Are Being Recalled Nationwide—Over 3 Million Bottles Affected
Eye Drops Are Being Recalled Nationwide—Over 3 Million Bottles Affected
The health industry is currently facing a massive safety crisis as a nationwide recall has been issued for over 3 million bottles of eye drops. This alert comes after federal regulators identified significant risks of bacterial contamination and lack of sterility in several popular over-the-counter brands. For millions of Americans who rely on these products for dry eye relief, allergy management, or daily lubrication, the news is both alarming and urgent.
The Food and Drug Administration (FDA) has stepped up its oversight, warning consumers that using these tainted products could lead to severe eye infections, permanent vision loss, or even life-threatening systemic complications. This recall is one of the largest in recent pharmaceutical history, affecting major retail chains and online marketplaces across the United States.
Understanding the Scope of the Massive Eye Drop Recall
The scale of this recall is unprecedented. When we talk about "3 million bottles," we are referring to a diverse range of products distributed under various labels. These include generic store brands found at major pharmacies like CVS, Rite Aid, and Target, as well as specific specialized brands often recommended by optometrists. The sheer volume of affected units suggests a systemic failure in manufacturing or quality control processes at the production level.
According to the FDA’s latest safety communication, the primary concern stems from "unsanitary conditions in the manufacturing facility." During recent inspections, officials discovered that the environments used to produce these sterile ophthalmic solutions were compromised. For a product that is applied directly to the surface of the eye—a highly sensitive organ with a direct pathway to the bloodstream—the requirement for absolute sterility is non-negotiable.
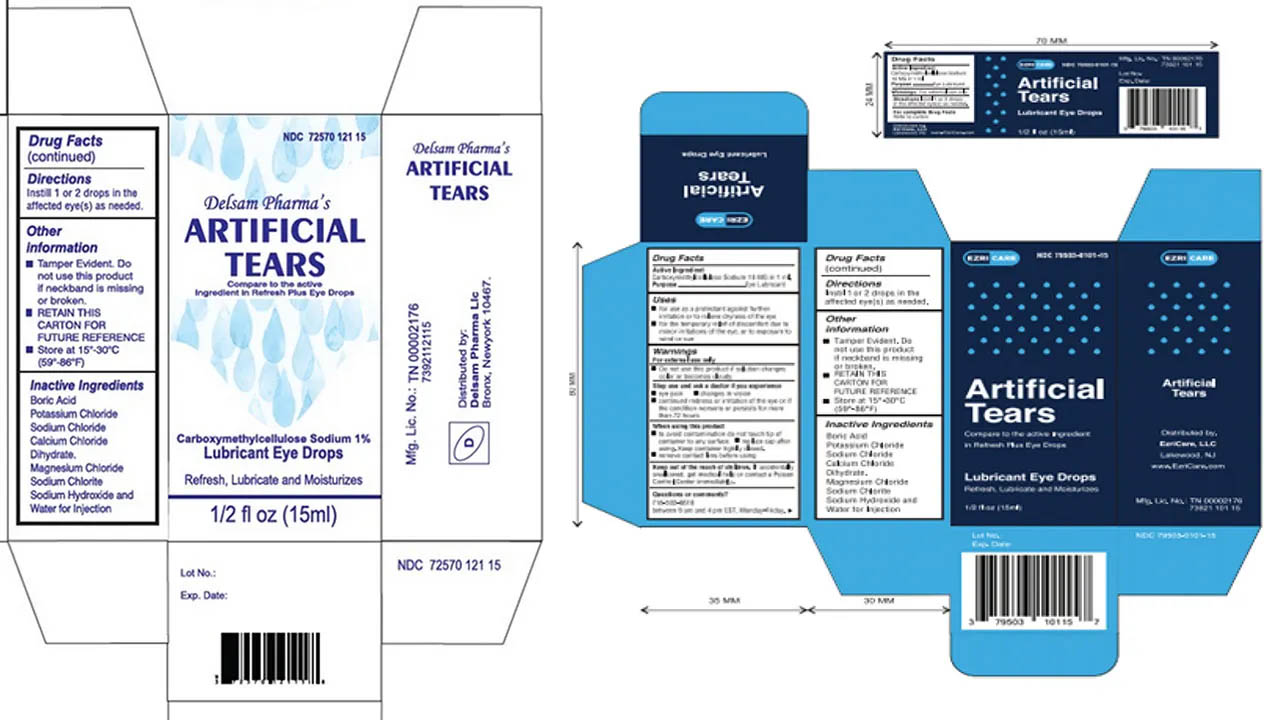
- AmniVisual and Dr. Berne’s: Some of the first brands to be flagged due to unauthorized ingredients and lack of sterility.
- Cardinal Health and Rugby Laboratories: Major distributors that have pulled thousands of units of lubricating drops.
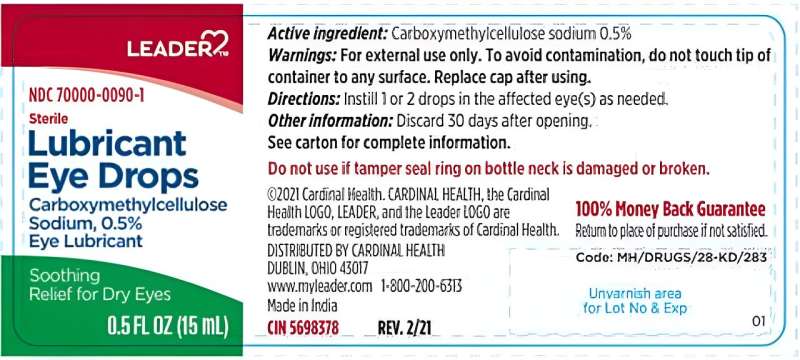
- Store Brands: Various "Equate" (Walmart), "Up & Up" (Target), and "Leader" (Cardinal Health) products are under scrutiny.
- Specialty Tears: Several types of carboxymethylcellulose sodium drops used for chronic dry eye.
The investigation reveals that many of these products were produced in facilities that failed to adhere to Current Good Manufacturing Practices (CGMP). In some instances, testers found traces of bacteria, including Pseudomonas aeruginosa, a resilient pathogen known for causing difficult-to-treat infections in humans. For consumers, this means that a simple act of soothing an itchy eye could inadvertently introduce a dangerous colony of bacteria into their system.
Why Sterility Matters: The Hidden Dangers of Contaminated Eye Care
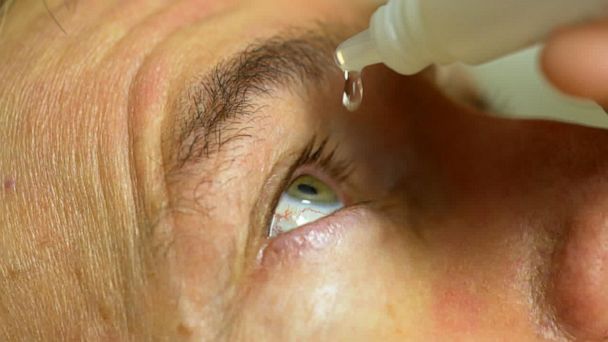
To understand why this recall is being treated with such gravity, one must understand the unique vulnerability of the human eye. Unlike the skin, which acts as a robust barrier against environmental pathogens, the ocular surface is delicate. The cornea and conjunctiva are susceptible to rapid infection if exposed to non-sterile substances.
When an eye drop bottle is contaminated during the manufacturing process, every single drop dispensed is a potential carrier for disease. If a user has a minor scratch on their cornea—common among contact lens wearers or those with dry eyes—the bacteria can enter the deeper tissues of the eye almost instantly. This can lead to a condition known as endophthalmitis, an inflammation of the interior of the eye that constitutes a true medical emergency.
The FDA’s warning emphasizes that contaminated eye drops are particularly dangerous because they bypass the body’s primary external defenses. Symptoms of an infection caused by tainted drops may include:
- Severe eye pain or redness.
- Increased sensitivity to light (photophobia).
- Blurry or hazy vision.
- Yellow, green, or clear discharge from the eye.
- The feeling of having something stuck in your eye.
If left untreated, these infections can progress rapidly. In previous, smaller recalls earlier this year, several individuals reported permanent blindness, and in extreme cases, the surgical removal of the eyeball was required to stop the spread of infection. This is why health officials are urging the public to stop using the affected products immediately, even if they have not yet experienced symptoms.
The Human Impact: A Story of Caution
To put a face to this technical alert, consider the story of Sarah, a 42-year-old graphic designer from Ohio. Sarah, like millions of others, spent long hours in front of a computer screen. To combat the inevitable eye strain and dryness, she kept a bottle of over-the-counter lubricating drops at her desk. She had used the same brand for years, trusting the "sterile" label on the packaging.
One Tuesday morning, Sarah noticed her left eye was slightly pink. She applied her usual drops, thinking it was just irritation. By Wednesday evening, her vision was cloudy, and the pain had become unbearable. "It felt like someone was pressing a hot needle into my pupil," she later recalled. After a late-night trip to the emergency room, she was diagnosed with a bacterial corneal ulcer.
Fortunately, Sarah’s doctors were able to identify the infection early, but she spent three weeks on intensive antibiotic therapy and missed nearly a month of work. Her bottle of eye drops was later identified as part of a batch that had been manufactured in a facility with poor sanitation. "I never thought something I bought at a reputable pharmacy to help my eyes would end up nearly blinding me," she said. Sarah's story is a cautionary tale for the 3 million other potential victims currently holding these bottles in their medicine cabinets.
Identifying Affected Products: What Consumers Need to Check Today
With so many brands involved, identifying whether your specific bottle is part of the recall can be confusing. The FDA and the manufacturing companies are working to provide specific lot numbers and expiration dates, but the general rule of thumb is "when in doubt, throw it out."
Most of the affected products are "Lubricant Eye Drops" or "Artificial Tears." These are generally used to relieve dryness caused by wind, sun, or computer use. Consumers should look at the brand name on the front of the bottle. If your drops are from any of the following retailers, check the official FDA recall list immediately:
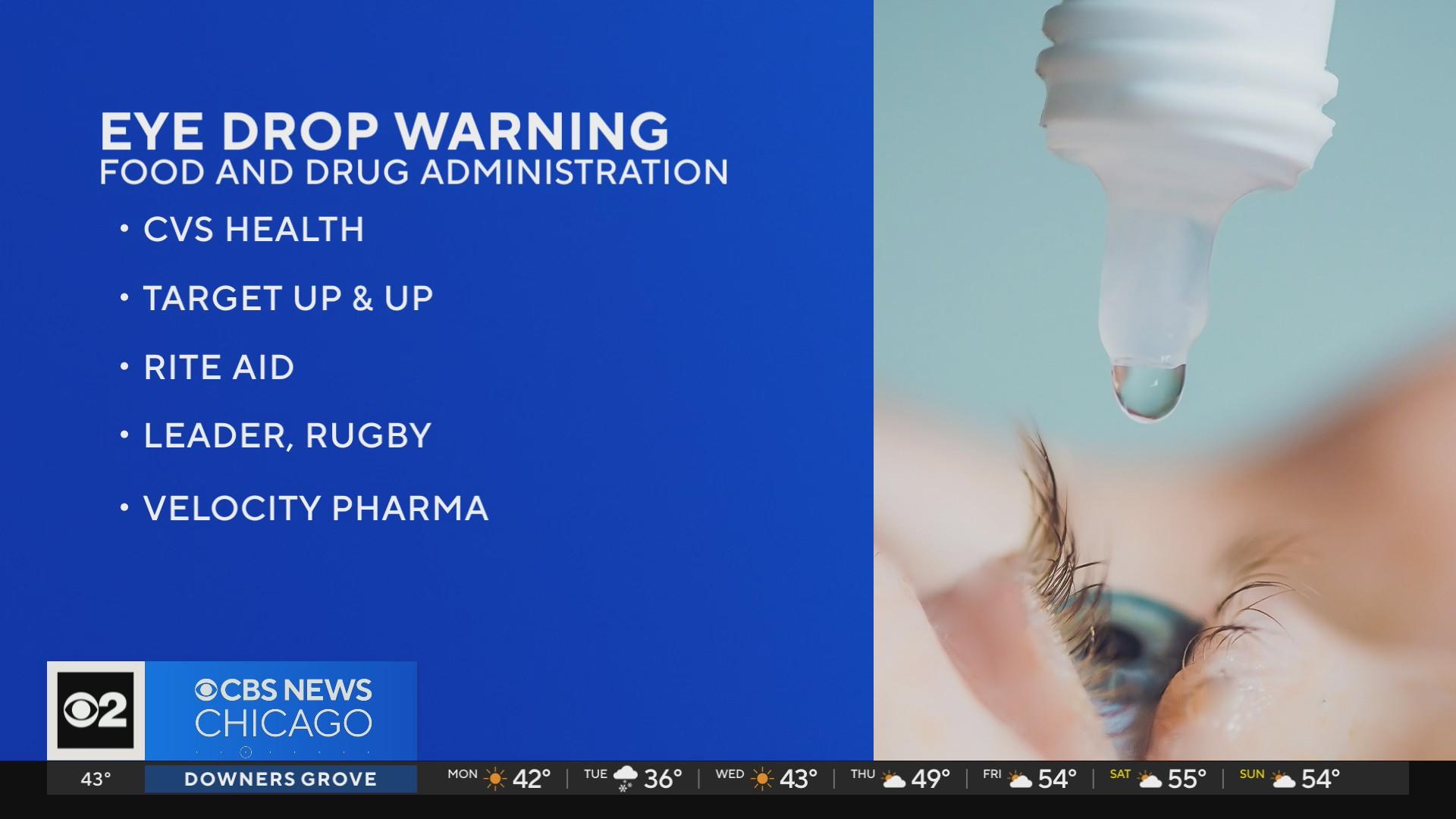
- CVS Health
- Rite Aid
- Target (Up & Up)
- Walmart (Equate)
- Velocity Pharma
- Leader (Cardinal Health)
- Rugby Laboratories
The FDA recommends that consumers do not simply throw these bottles in the trash where children or pets might find them. Instead, follow the disposal instructions provided on the recall notice, or return them to the place of purchase for a refund. Many retailers have already removed these items from their shelves, but the millions of bottles already in circulation remain a primary concern for public health officials.
Furthermore, this recall highlights a significant loophole in how over-the-counter eye care is regulated. Unlike prescription drugs, which undergo rigorous pre-market approval, many OTC drops are marketed based on "monographs"—sets of rules that allow companies to sell products without individual FDA approval as long as they follow established guidelines. When companies cut corners on manufacturing safety to save costs, the results can be catastrophic.
Step-by-Step Guide: What to Do If You Have Recalled Eye Drops
If you discover that your eye drops are part of the nationwide recall, do not panic, but act swiftly. Your priority should be preventing further exposure and monitoring your health.
1. Stop Use Immediately: Even if you have used the drops recently and feel fine, stop immediately. Bacterial contamination is not always visible to the naked eye; the solution may look perfectly clear despite being loaded with pathogens.
2. Check for Symptoms: Monitor your eyes for the next 48 to 72 hours. Look for redness, discharge, or any changes in your vision. If you wear contact lenses, pay extra attention to any discomfort.
3. Consult a Professional: If you experience any symptoms, contact an ophthalmologist or optometrist right away. Tell them specifically that you have been using a recalled eye drop product. This information is vital for them to choose the correct antibiotic treatment.
4. Preserve the Product: If you are already experiencing symptoms, do not throw the bottle away yet. Keep it in a sealed plastic bag. Your doctor or health authorities may want to test the liquid to identify the specific strain of bacteria causing your infection.
5. Report to the FDA: Consumers and healthcare professionals are encouraged to report any adverse events or quality problems with these products to the FDA’s MedWatch Adverse Event Reporting program. This helps the government track the spread of the issue and hold companies accountable.
Looking Ahead: How to Ensure Your Future Eye Health Safety
This massive recall serves as a wake-up call for both the pharmaceutical industry and consumers. While we often take the safety of over-the-counter products for granted, this event proves that vigilance is necessary. Moving forward, there are several steps you can take to ensure the products you use are safe.
First, always check for the "sterile" label on any eye care product. While the recalled products had this label, it is still a basic requirement for any ophthalmic solution. Second, stay informed about FDA alerts. You can sign up for email notifications from the FDA regarding drug recalls and safety communications.
Third, consider the packaging. Some newer eye drop designs use multi-dose preservative-free (MDPF) bottles that have built-in valves to prevent bacteria from entering the bottle after it has been opened. While these are more expensive, they offer an extra layer of protection for those with sensitive eyes.
Finally, support legislative efforts for stricter oversight. The current crisis has prompted calls for the FDA to have more authority to inspect foreign and domestic manufacturing plants more frequently. Ensuring that every bottle produced meets the highest safety standards is the only way to prevent a recall of this magnitude from happening again.
In conclusion, the recall of over 3 million bottles of eye drops is a serious health event that requires immediate action from consumers nationwide. By checking your medicine cabinet, staying informed, and seeking medical help at the first sign of trouble, you can protect your vision and your health from the dangers of contaminated eye care. Stay safe, stay vigilant, and always prioritize your ocular health in an increasingly complex pharmaceutical landscape.
Eye Drops Are Being Recalled Nationwide—Over 3 Million Bottles Affected
Eye Drops Are Being Recalled Nationwide—Over 3 Million Bottles Affected Wallpapers
Collection of eye drops are being recalled nationwide—over 3 million bottles affected wallpapers for your desktop and mobile devices.

Mesmerizing Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Photo Photography
Transform your screen with this vivid eye drops are being recalled nationwide—over 3 million bottles affected artwork, a true masterpiece of digital design.
.png)
Vivid Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Scene Photography
Explore this high-quality eye drops are being recalled nationwide—over 3 million bottles affected image, perfect for enhancing your desktop or mobile wallpaper.
Vivid Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Artwork Illustration
Experience the crisp clarity of this stunning eye drops are being recalled nationwide—over 3 million bottles affected image, available in high resolution for all your screens.

Breathtaking Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Image Nature
Discover an amazing eye drops are being recalled nationwide—over 3 million bottles affected background image, ideal for personalizing your devices with vibrant colors and intricate designs.

Lush Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Moment Collection
Find inspiration with this unique eye drops are being recalled nationwide—over 3 million bottles affected illustration, crafted to provide a fresh look for your background.
Lush Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Image Art
Immerse yourself in the stunning details of this beautiful eye drops are being recalled nationwide—over 3 million bottles affected wallpaper, designed for a captivating visual experience.

Breathtaking Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Landscape for Your Screen
Transform your screen with this vivid eye drops are being recalled nationwide—over 3 million bottles affected artwork, a true masterpiece of digital design.

Detailed Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Scene Concept
Transform your screen with this vivid eye drops are being recalled nationwide—over 3 million bottles affected artwork, a true masterpiece of digital design.

Mesmerizing Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Image for Mobile
Experience the crisp clarity of this stunning eye drops are being recalled nationwide—over 3 million bottles affected image, available in high resolution for all your screens.

Spectacular Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Capture Concept
A captivating eye drops are being recalled nationwide—over 3 million bottles affected scene that brings tranquility and beauty to any device.

Captivating Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Picture Digital Art
A captivating eye drops are being recalled nationwide—over 3 million bottles affected scene that brings tranquility and beauty to any device.

Gorgeous Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Design in 4K
A captivating eye drops are being recalled nationwide—over 3 million bottles affected scene that brings tranquility and beauty to any device.

Crisp Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Artwork in 4K
This gorgeous eye drops are being recalled nationwide—over 3 million bottles affected photo offers a breathtaking view, making it a perfect choice for your next wallpaper.

Beautiful Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Artwork in 4K
Discover an amazing eye drops are being recalled nationwide—over 3 million bottles affected background image, ideal for personalizing your devices with vibrant colors and intricate designs.

Serene Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Design in 4K
Experience the crisp clarity of this stunning eye drops are being recalled nationwide—over 3 million bottles affected image, available in high resolution for all your screens.

Dynamic Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Capture Collection
This gorgeous eye drops are being recalled nationwide—over 3 million bottles affected photo offers a breathtaking view, making it a perfect choice for your next wallpaper.

Amazing Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Abstract Collection
Experience the crisp clarity of this stunning eye drops are being recalled nationwide—over 3 million bottles affected image, available in high resolution for all your screens.
Vivid Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Background for Your Screen
Transform your screen with this vivid eye drops are being recalled nationwide—over 3 million bottles affected artwork, a true masterpiece of digital design.

Artistic Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Landscape in HD
A captivating eye drops are being recalled nationwide—over 3 million bottles affected scene that brings tranquility and beauty to any device.
Exquisite Eye Drops Are Being Recalled Nationwide—over 3 Million Bottles Affected Landscape in 4K
Immerse yourself in the stunning details of this beautiful eye drops are being recalled nationwide—over 3 million bottles affected wallpaper, designed for a captivating visual experience.
Download these eye drops are being recalled nationwide—over 3 million bottles affected wallpapers for free and use them on your desktop or mobile devices.
0 Response to "Eye Drops Are Being Recalled Nationwide—Over 3 Million Bottles Affected"
Post a Comment